SARS-CoV-2 omicron XBB.1.5 variant is highly transmissible and infectious, study shows
COVID-19 has caused significant global panic after its rapid emergence more than three years ago. Although we now have highly effective vaccines against the SARS-CoV-2 virus, which causes COVID-19, scientists continue to study emerging SARS-CoV-2 variants in order to safeguard public health and devise global preventive strategies against emerging variants. A team led by Japanese researchers has recently discovered that the SARS-CoV-2 omicron XBB.1.5 variant, prevalent in the Western hemisphere, has high transmissibility and infectivity.
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has been responsible for millions of deaths worldwide. Although scientists have designed novel vaccines to counter COVID-19, they are constantly on the lookout for emerging variants that can bypass vaccine resistance and potentially jeopardize global public health. A team led by Japanese researchers has recently been successful in characterizing the new SARS-CoV-2 omicron XBB.1.5 variant, which was first detected in October 2022. Their findings were published in The Lancet Infectious Diseases.
Senior author Prof. Kei Sato from the Division of Systems Virology, The Institute of Medical Science, The University of Tokyo, Japan, says, “Because the omicron XBB.1.5 variant can spread more rapidly than previous variants and has a potential to cause the next epidemic surge, we should carefully monitor it to safeguard public health.”
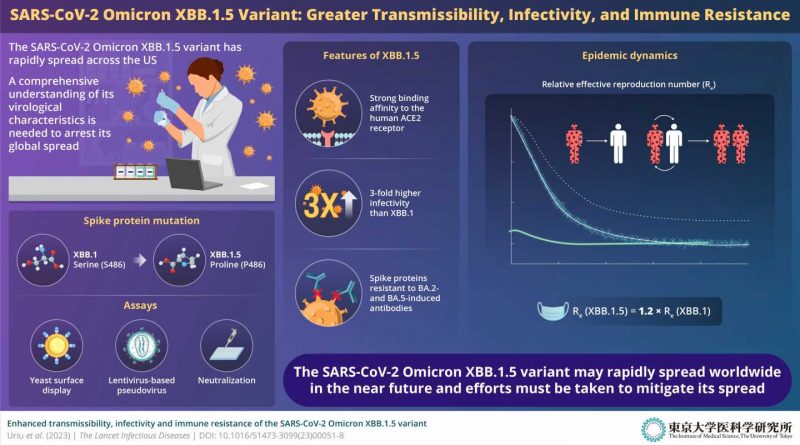
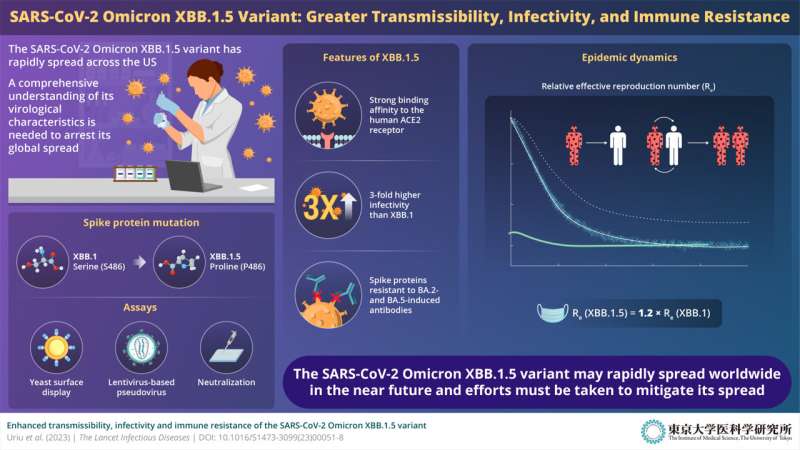
While studying emerging variants of the SARS-CoV-2 omicron lineage, the research team made a startling discovery: the SARS-CoV-2 omicron XBB.1.5 variant has a novel mutation in the spike (S) protein, which is the protein that anchors the virus firmly to the human angiotensin converting enzyme-2 (ACE2) receptor, thus facilitating the invasion of human cells. The serine-to-proline amino acid mutation noted at residue no. 486 in the S protein is virologically concerning because of a variety of reasons.
Sharing his concerns, first author Keiya Uriu from the Division of Systems Virology, Department of Microbiology and Immunology, The University of Tokyo, Japan, says, “In late 2022, the SARS-CoV-2 omicron BQ.1 and XBB lineages, characterized by amino acid substitutions in the S protein and increased viral fitness, had become predominant in the Western and Eastern Hemisphere, respectively. In 2022, we elucidated the characteristics of a variety of newly emerging SARS-CoV-2 omicron subvariants. At the end of 2022, the XBB.1.5 variant, a descendant of XBB.1 that acquired the S:S486P substitution, emerged and was rapidly spreading in the U.S.”
To gain mechanistic insights into the infectivity, transmissibility, and immune response associated with XBB.1.5, the team conducted a series of experiments. For instance, upon conducting epidemic dynamics analysis—statistical modeling that facilitates the analysis of the general characteristics of any epidemic—the team realized that the relative effective reproduction number (Re) of XBB.1.5 was 1.2-fold greater than that of the parental XBB.1. This indicated that an individual with the XBB.1.5 variant could infect 1.2 times more people in the population than someone with the parental XBB.1 variant. Moreover, the team also realized that, as of December 2022, XBB.1.5 was rapidly outcompeting BQ.1.1, the predominant lineage in the United States.
Co-first-author Jumpei Ito from the Division of Systems Virology, says, “Our data suggest that XBB.1.5 will rapidly spread worldwide in the near future.”
The team also studied the virological features of XBB.1.5 to determine how tightly the S protein of the new variant interacts with the human ACE2 receptor. To this end, the researchers conducted a yeast surface display assay. The results showed that the dissociation constant (KD) corresponding to the physical interaction between the XBB.1.5 S receptor-binding domain (RBD) and the human ACE2 receptor is significantly (4.3-fold) lower than that for XBB.1 S RBD. “In other words, the XBB.1.5 variant binds to human ACE2 receptor with very high affinity,” explains Shigeru Fujita from the Division of Systems Virology.
Further experiments using lentivirus-based pseudoviruses also showed that XBB.1.5 had approximately 3-fold higher infectivity than XBB.1. These results suggest that XBB.1.5 exhibits a remarkably strong affinity to the human ACE2 receptor, which can be attributed to the S486P substitution.
The study by Prof. Sato and his team led to another important discovery from an immunization perspective. The XBB.1.5 S protein was found to be highly resistant to neutralization antibodies elicited by breakthrough infection with the BA.2/BA.5 subvariants. In other words, patients with prior infection from the BA.2/BA.5 subvariants may not show robust immunity against XBB.1.5, increasing their chances of infection and disease.
“The results of our virological experiments explain why the omicron XBB.1.5 variant has a higher transmissibility than past variants: This variant acquired strong binding ability to human ACE2 while maintaining a higher ability to escape from neutralizing antibodies,” says Yusuke Kosugi from the Division of Systems Virology, Department of Microbiology and Immunology.
Contributing members of The Genotype to Phenotype Japan (G2P-Japan) Consortium conclude, “The SARS-CoV-2 omicron XBB.1.5 variant does show enhanced transmissibility. Although few cases have been detected in the Eastern hemisphere, it could become a looming threat. Imminent prevention measures are needed.”
More information:
Keiya Uriu et al, Enhanced transmissibility, infectivity, and immune resistance of the SARS-CoV-2 omicron XBB.1.5 variant, The Lancet Infectious Diseases (2023). DOI: 10.1016/S1473-3099(23)00051-8
Journal information:
Lancet Infectious Diseases
Source: Read Full Article